Cure4CF Update
Thinking About Joining a Clinical Trial? Here’s What You Should Know
Professor Jodie Simpson, Head of Research, Cure4CF
I was recently invited to take part in a clinical trial, and it made me stop and think about how many people might not really know what that means. Clinical trials are often mentioned in the news or by health professionals, but what happens in one—and why do people volunteer?
Let’s unpack it a bit.
What is a clinical trial?
Clinical trials are how we test new treatments and ideas in medicine. They’re a way to find out whether something new—a medicine, a medical device, or even a way of delivering a treatment—really works and is safe. Sometimes it’s about testing a brand-new treatment. Other times, it’s comparing two existing options to see which is better, or trying a new way of diagnosing or managing a condition.
Trials can even look at how health services are delivered to see if one approach works better for patients than another. Whatever the focus, clinical trials are a vital part of improving healthcare and, ultimately, improving lives.
What about the different phases?
You might have heard of Phase 1, 2 or 3 trials. In simple terms, these are just stages in the testing process. Even treatments in early-phase trials have already gone through extensive safety testing—often starting in healthy volunteers before involving people with the condition the treatment is meant for. By the time a treatment reaches Phase 3, researchers usually already have a good idea that it’s safe, and they’re testing how well it works compared to standard treatments.
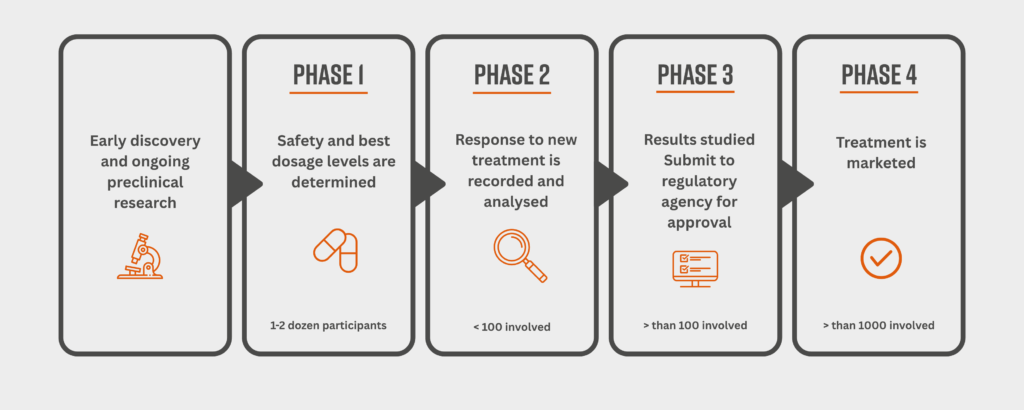
Why are clinical trials so important?
Clinical trials are how we make sure that new treatments, devices, and models of care are both safe and effective. Without them, medicine would never move forward. Participating in one means you’re part of something that could genuinely change future healthcare—for yourself and for others.
What’s it actually like to be in a trial?
I’ve been in quite a few over the years! I’ve done everything from flu vaccine studies (involving nasal washes every few months) to being a healthy control in asthma and COPD studies—lots of breathing tests and sputum samples. I even took part in an asthma and pregnancy study and a study where they used a tiny camera to look at my vocal cords. That one was fascinating!
For me, every study has been a chance to learn something new—not just about science, but about myself. I’m naturally curious, so I’ve loved the experience of contributing to health and science and meeting the teams who make these studies happen.
When I first started, I was nervous about blood tests (my veins are a bit hopeless), so I’d always ask if that was part of the study before signing up and often I could participate in the study and just not do some of the tests. These days, after having two kids, I’m much better with needles!
How do you decide if a trial is right for you?
It’s a personal choice, and it’s okay to take your time. Talk it over with your healthcare team, and maybe a trusted family member or friend. Ask questions—lots of them! You’ll want to know what the study hopes to learn, whether there’s a placebo (a dummy medicine), and what kind of commitment is involved. Don’t be afraid of being in a study with a placebo, yes you might get the placebo, but it is crucial to knowing if a medicine works!
And remember: you can ask the research team questions at any stage—before, during, or after you join.
Can you change your mind?
Absolutely. You can withdraw from a trial at any time, for any reason. It’s your choice, and the research team will always respect that. If you don’t feel that you have been respected, then on the information and consent form you will find the contact details of the ethics committee that approved the study where you can express your concerns. The ethics committee’s I have worked with take this really seriously and work with teams to ensure that people feel safe.
What are the advantages of taking part?
There are plenty. You might get early access to a treatment that isn’t yet available to the public. You’ll also be contributing to scientific knowledge and helping others who may benefit from what researchers learn. Plus, being in a trial often means you’re seen more regularly by healthcare professionals, which can give you extra insight into your condition.
And the downsides?
Trials can take up a bit more of your time—you might need extra appointments, tests, or even fill out more paperwork. Sometimes there are side effects from the treatment being tested, but you’ll be told everything that’s known about the risks before you agree to take part, and the team will monitor you closely. In one of the clinical trial’s I ran headache was a common side effect – but it was experienced just as much in those who had the treatment we were testing as in the group who received the placebo treatment. Another reason the placebo group is vital – it helps to know if side effects are from the treatment itself or things that are occurring randomly.
The bottom line
Clinical trials are one of the most powerful tools we have for improving health and healthcare. If you’re thinking about joining one, learn as much as you can, ask all your questions, and make a decision that feels right for you.
And who knows—you might just find, like I did, that it’s a fascinating and rewarding experience.
